Press release
New Data Presented from Oncopeptides’ Pivotal Phase 2 HORIZON Trial Evaluating Melflufen in Relapsed/Refractory Multiple Myeloma at 24th EHA Congress
The presentation will be available on the company webpage under:
www.oncopeptides.com / Investors & media / Presentations / 2019 EHA
Comment from CEO Jakob Lindberg
“HORIZON is an all-comer trial for patients with very advanced multiple myeloma. A majority of patients have extra medullary disease (EMD) as well as high-risk cytogenetics. Notably patients with EMD at relapse have limited response from treatment in clinical trials even in the era of modern therapies. In light of this patient population, the response rate of 28% coupled with the manageable side effect profile in HORIZON is very encouraging and indicates that melflufen plus dexamethasone can offer these patients a reasonable treatment alternative. These data form the basis of the upcoming New Drug Application (NDA) submission that we are currently preparing for accelerated approval of melflufen for the treatment of patients with triple-class refractory multiple myeloma” said Jakob Lindberg, CEO of Oncopeptides.
Overall Conclusions – HORIZON Presentation
- At the time for the data cut-off, May 6th, 35 out of the 121 patients included were still on treatment.
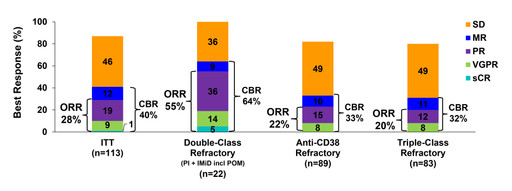
- The Overall Response Rate (ORR) was 28% and the Clinical Benefit Rate (CBR) was 40%, 36% of patients had receved 3+ treatment regimens over the last 12 months.
- Extra-medullary disease patients had an ORR of 29%.
- The majority of patients 86%, achieved disease stabilization (SD or better)
- The median Progression Free Survival (PFS) was 4.0 months in the ongoing study.
- Treatment was generally well tolerated with manageable toxicity, nonhematologic AEs were infrequent and the rate of discontinuation due to AEs was low.
Comment from Professor Paul G. Richardson
“The number of patients with resistant myeloma characterized by extra-medullary disease and high-risk cytogenetics is increasing, despite recent advances, and there is a real need for additional treatment options based on new mechanisms of action. Melflufen continues to demonstrate promising activity and manageable toxicity in the relapsed and refractory setting, and in particular amongst patients who are triple-class refractory ” said Paul Richardson, MD, the RJ Corman Professor of Medicine at Harvard Medical School and Director of Clinical Research at the Jerome Lipper Multiple Myeloma Center, Dana-Farber Cancer Institute, Boston, USA.
About the OP-106 HORIZON study
Patient recruitment in the HORIZON study is ongoing. The interim data presented at EHA is based on a data cut-off dated May 6th 2019 with 121 patients treated. 108 patients had recivied two or more cycles of treatment. The goal is to include 150 patients in the study. The patients in the study are refractory to pomalidomide and/or daratumumab after failing on immunomodulatory drugs (IMiDs) and proteasome inhibitors (PIs).
More information can be found at: https://clinicaltrials.gov/ct2/show/NCT02963493?term=melflufen&rank=2
Summary of the OP-106 HORIZON interim data
The study continues to develop positively in this heavily pretreated patient group. Melflufen continues to demonstrate promising activity in patients that have stopped responding to lenalidomide and PI based regimens and subsequently become resistant to salvage therapy based on daratumumab and/or pomalidomide.
The median age of the patients was 64 years. 62% of patients in the study had high-risk cytogenetics, 29% of patients were ISS stage III and 60% of the patients had extramedullary disease (EMD). The median number of prior lines of therapy was five and the median time since initial diagnosis was 6.2 years.
All patients in the study were investigator assessed as non-responsive or non-tolerant to IMiDs and PIs.
91% of patients were double class refractory (IMiD + PI) and 79% anti-CD38 mAb refractory. 74% of the patients were triple class (IMiD + PI + Anti-CD38 mAb) refractory and 59% were alkylator refractory. 98% of the patients were refractory in last line of therapy.
Efficacy
Analysis of the interim efficacy results showed an ORR of 28% and that 86% of the patients achieved disease stabilization (SD or better), see table below.

8 pts did not have available response information at data cutoff; 2 pts response evaluable, PI exposed, but refractoriness to PI subject to confirmation, so excluded from subgroup analysis
One pt with sCR also confirmed as MRD negative (10-6 sensitivity), with ongoing progression-free period of 13.6 mos
Median time to response 1.2 mos
CBR, clinical benefit rate; CR, complete response; MR, minimal response; ORR, overall response rate;
PD, progressive disease; PR, partial response; sCR, stringent CR; SD, stable disease; VGPR, very good PR.
For patients with extra-medullary disease the ORR was 29% as described in the table below.

Safety and tolerability
At time of data cut-off, treatment-related SAEs occurred in 20% of patients, most commonly febrile neutropenia (5%) and thrombocytopenia (2%). The overall incidence of nonhematologic AEs was low.
Discontinuation rate because of AEs was 20%. There were no treatment-related deaths. Six patients (6%) experienced treatment-related bleeding: grade 1 in four patients, grade 3 in two patients.
For further information, please contact:
Jakob Lindberg, CEO of Oncopeptides
E-mail: jakob.lindberg@oncopeptides.com
Telephone: +46 8 615 20 40
Rein Piir, Head of Investor Relations at Oncopeptides
E-mail: rein.piir@oncopeptides.com
Cell phone: +46 70 853 72 92
The information in the press release is information that Oncopeptides is obliged to make public pursuant to the EU Market Abuse Regulation. The information was submitted for publication, through the agency of the contact person above, on June 16, 2019 at 08.45 (CET).
About melflufen
Melflufen is a lipophilic peptide-conjugated alkylator that rapidly delivers a highly cytotoxic payload into myeloma cells through peptidase activity. It belongs to the novel class Peptidase Enhanced Cytotoxics (PEnC), which is a family of lipophilic peptides that exhibit increased activity via peptidase cleavage and have the potential to treat many cancers. Peptidases play a key role in protein homeostasis and feature in cellular processes such as cell-cycle progression and programmed cell death. Melflufen is rapidly taken up by myeloma cells due to its high lipophilicity and immediately cleaved by peptidases to deliver an entrapped hydrophilic alkylator payload. In vitro, melflufen is 50-fold more potent in myeloma cells than the alkylator payload itself due to the peptidase cleavage, and induces irreversible DNA damage and apoptosis. Melflufen displays cytotoxic activity against myeloma cell lines resistant to other treatments, including alkylators, and has also demonstrated inhibition of DNA repair induction and angiogenesis in preclinical studies.
About Oncopeptides
Oncopeptides is a pharmaceutical company focused on the development of targeted therapies for difficult-to-treat hematological cancers. The company is focusing on the development of the lead product candidate melflufen, a novel lipophilic peptide conjugated alkylator, belonging to a new class of drugs called Peptidase Enhanced Cytotoxics (PEnC). Melflufen is in development as a new treatment for the hematological cancer multiple myeloma, including the Phase 2 pivotal trial HORIZON currently underway and a global confirmatory Phase 3 trial (OCEAN) continuing enrollment. Oncopeptides’ headquarters is located in Stockholm, Sweden, and the company is listed in the Mid Cap segment on Nasdaq Stockholm with the ticker ONCO.